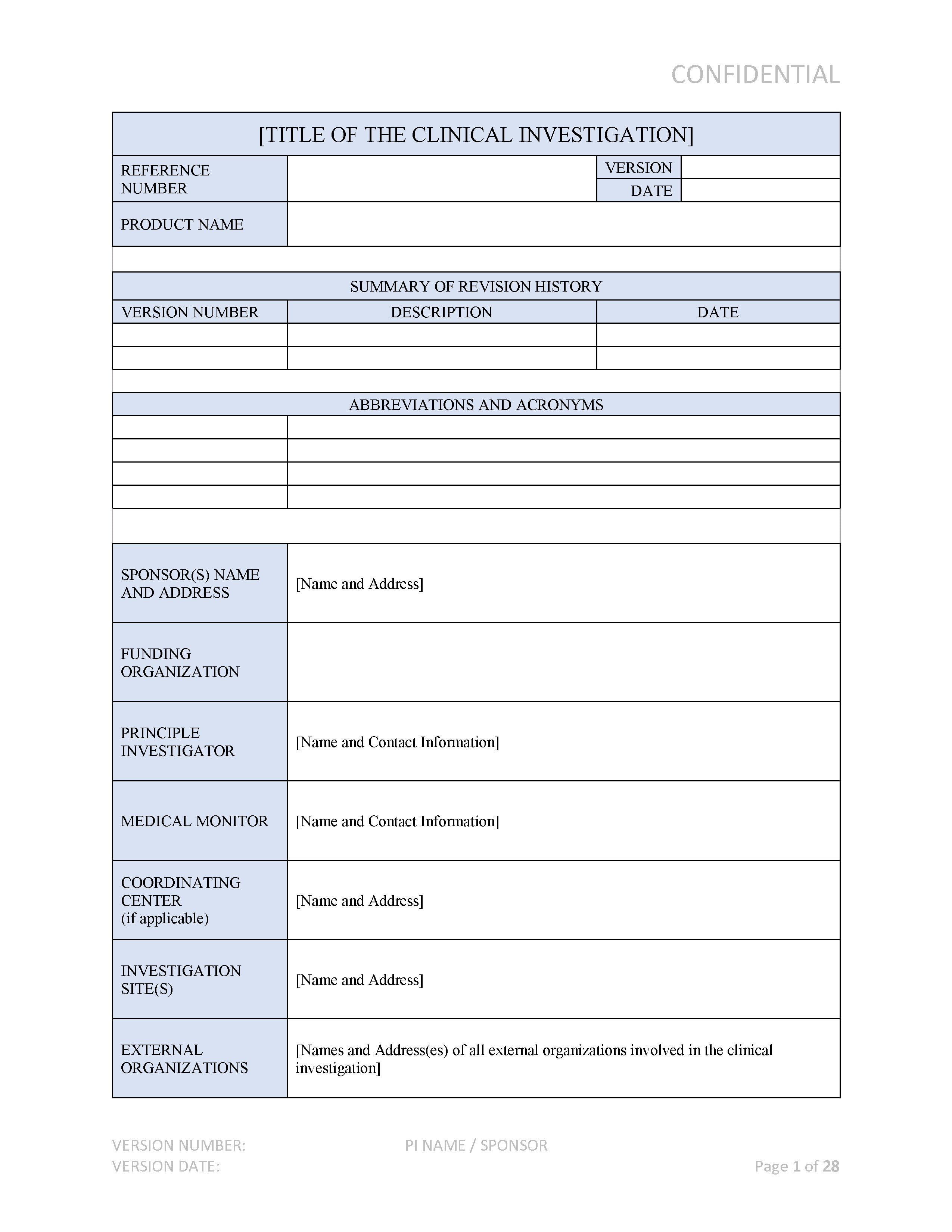
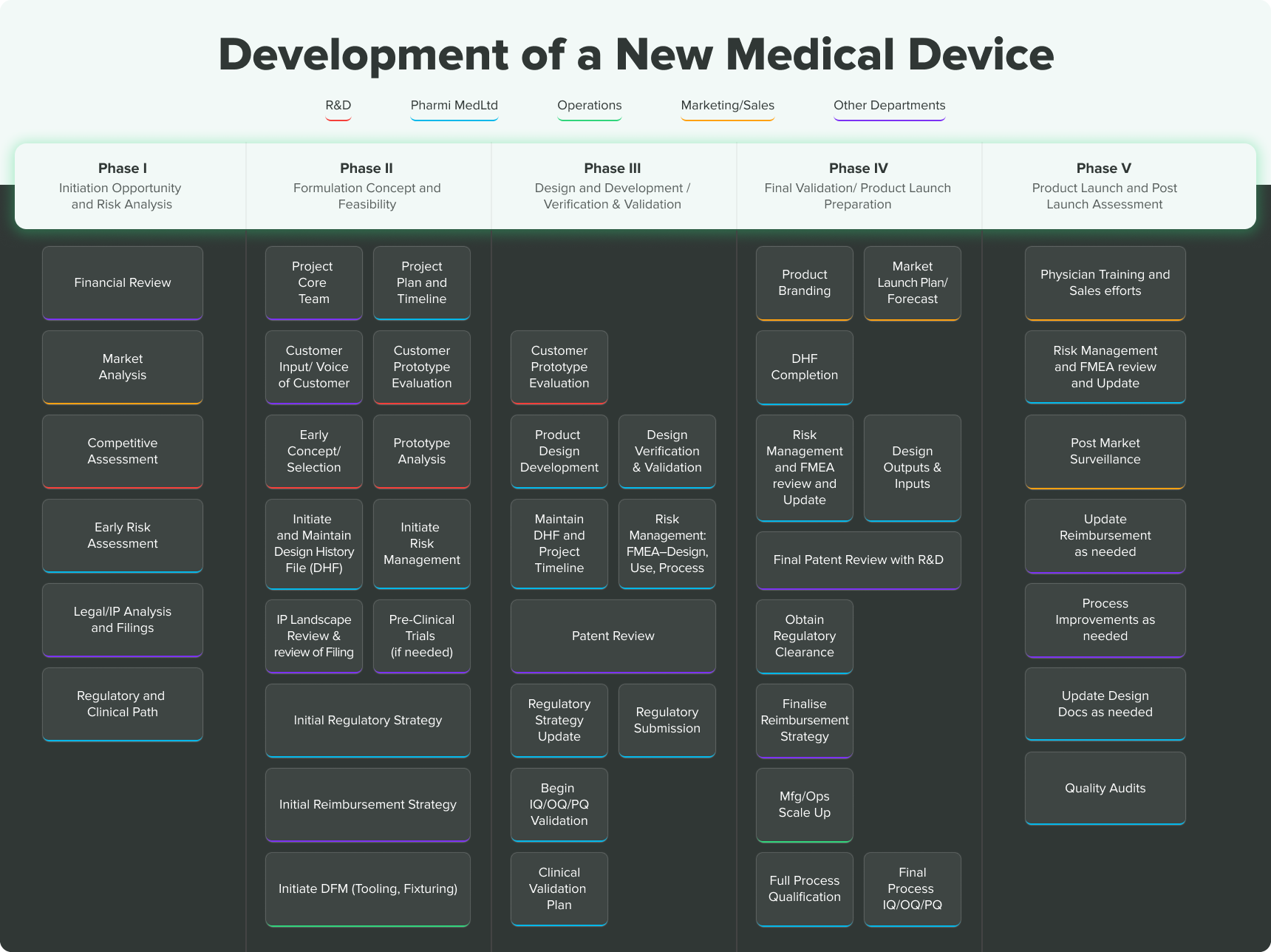
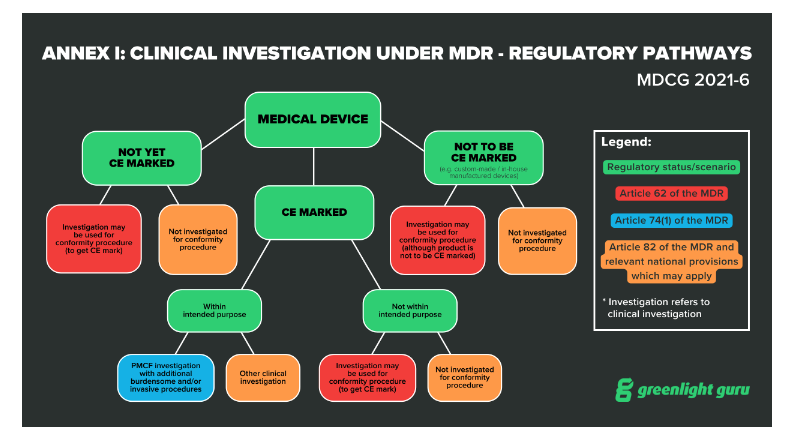
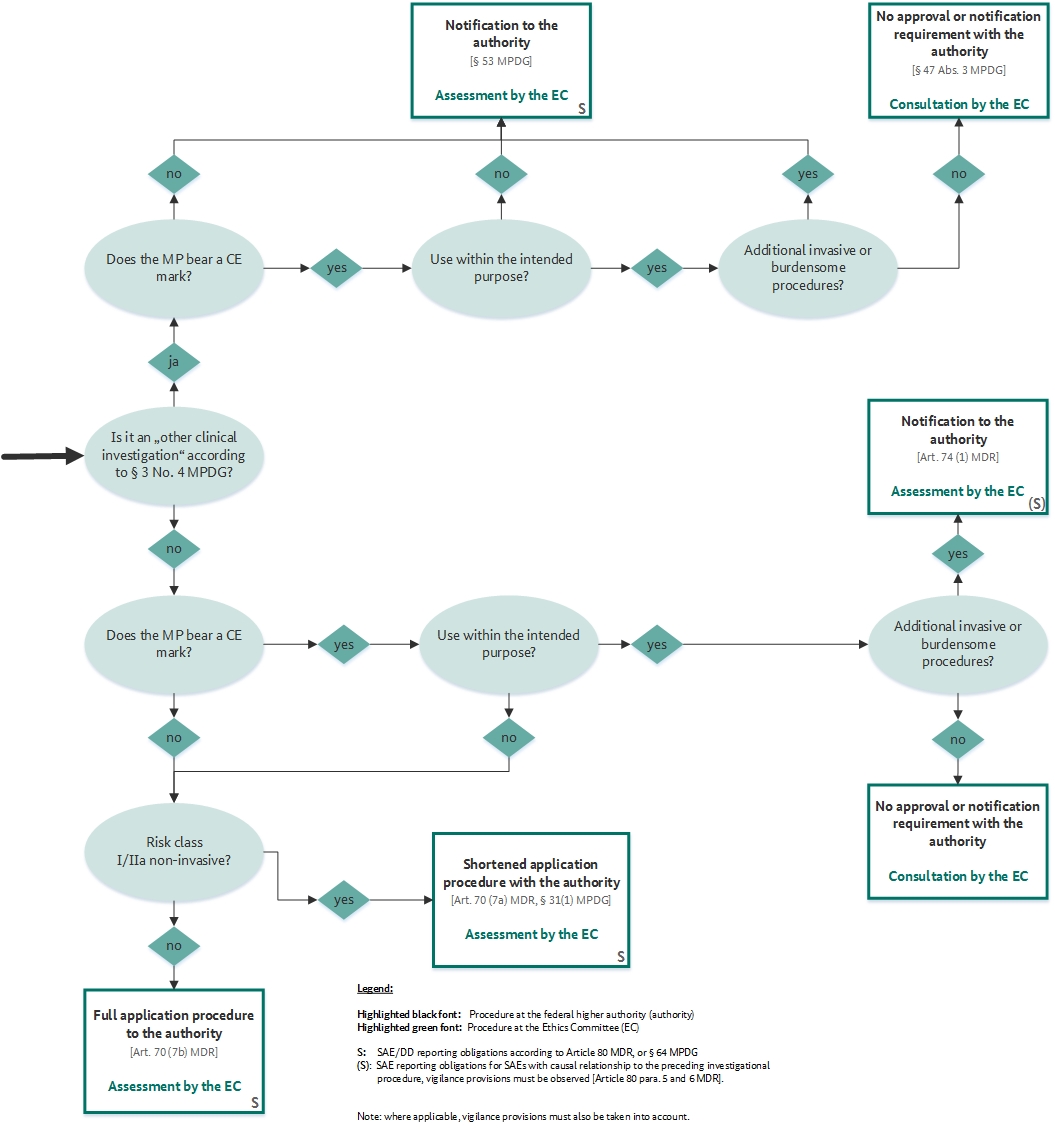
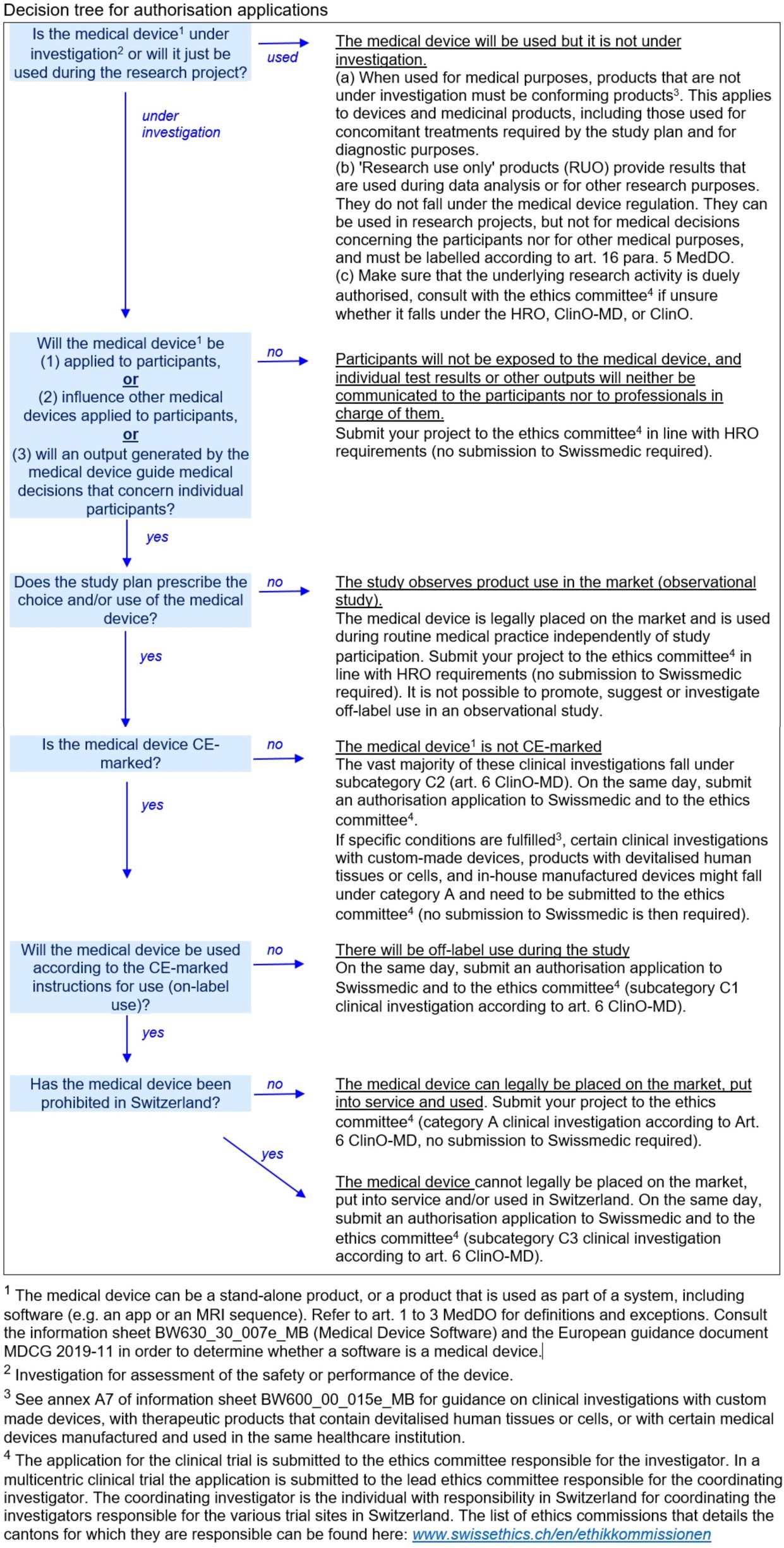
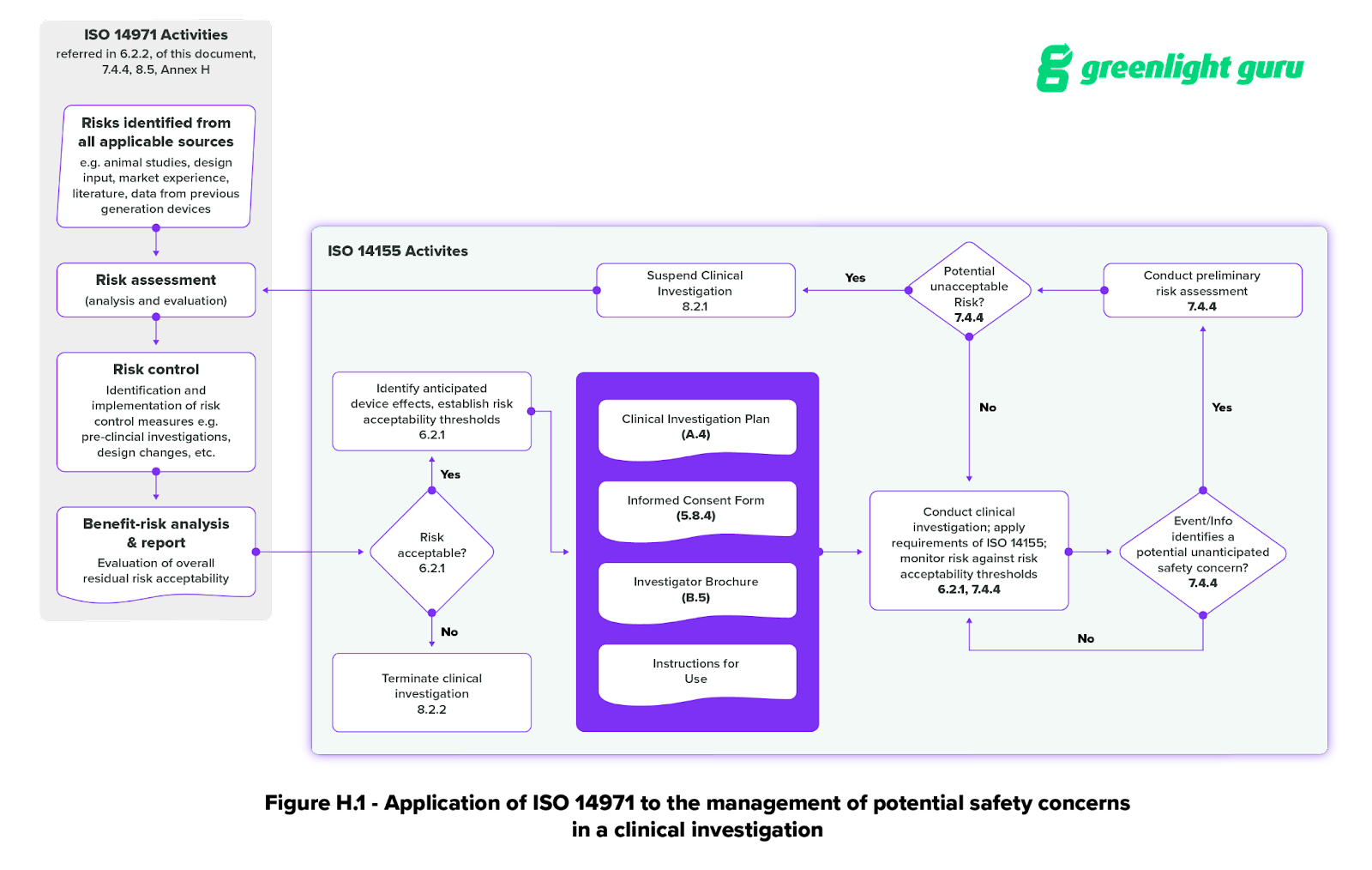
GUIDELINES ON MEDICAL DEVICES GUIDELINES FOR COMPETENT AUTHORITIES FOR MAKING A VALIDATION/ASSESSMENT OF A CLINICAL INVESTIGATIO
Evolving regulatory perspectives on digital health technologies for medicinal product development | npj Digital Medicine

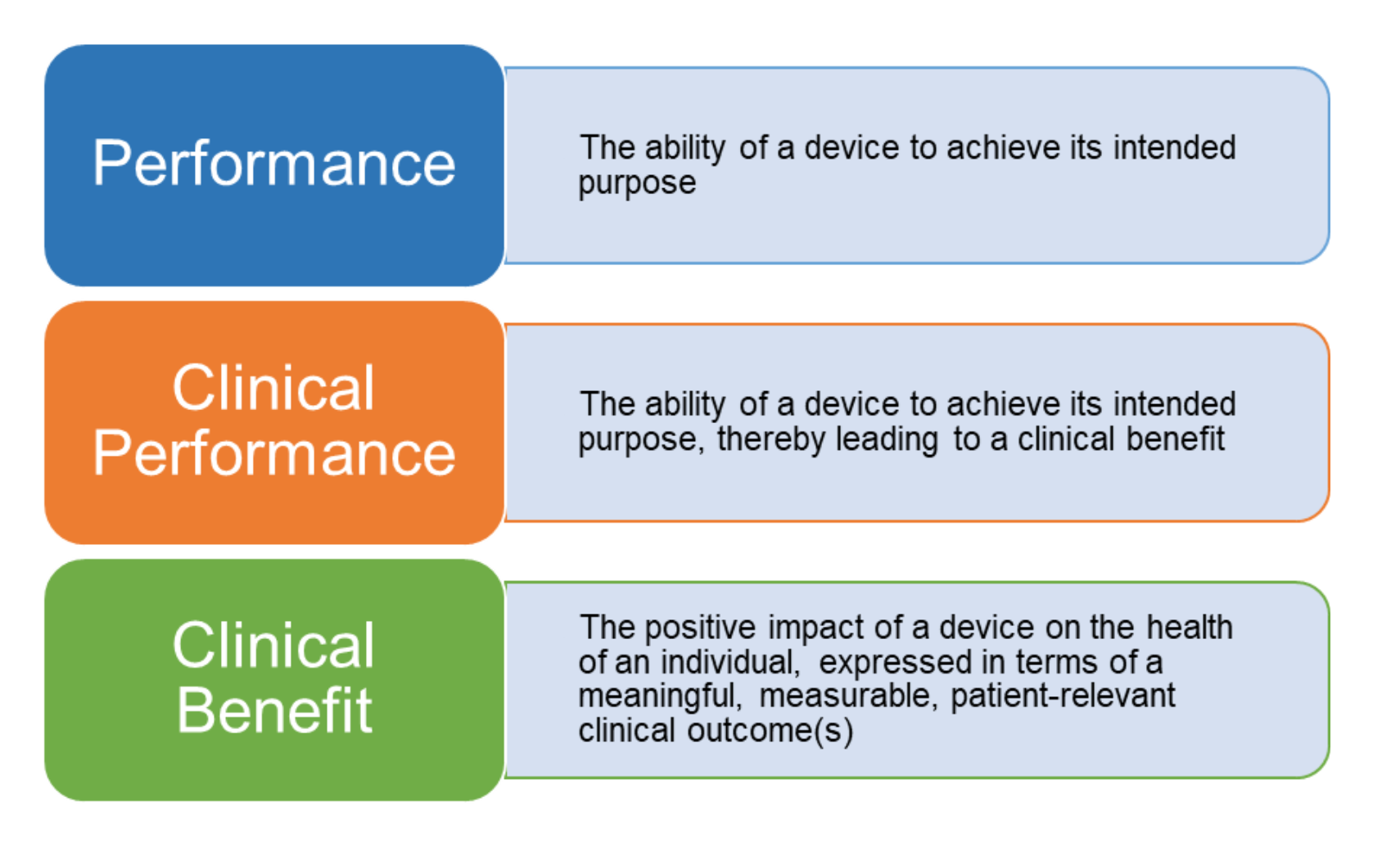
Improved clinical investigation and evaluation of high-risk medical devices: the rationale and objectives of CORE–MD (Coordinating Research and Evidence for Medical Devices) in: EFORT Open Reviews Volume 6 Issue 10 (2021)

ANSI/AAMI/ISO 14155-2:2003 - Clinical investigation of medical devices for human subjects - Part 2: Clinical investigation plans