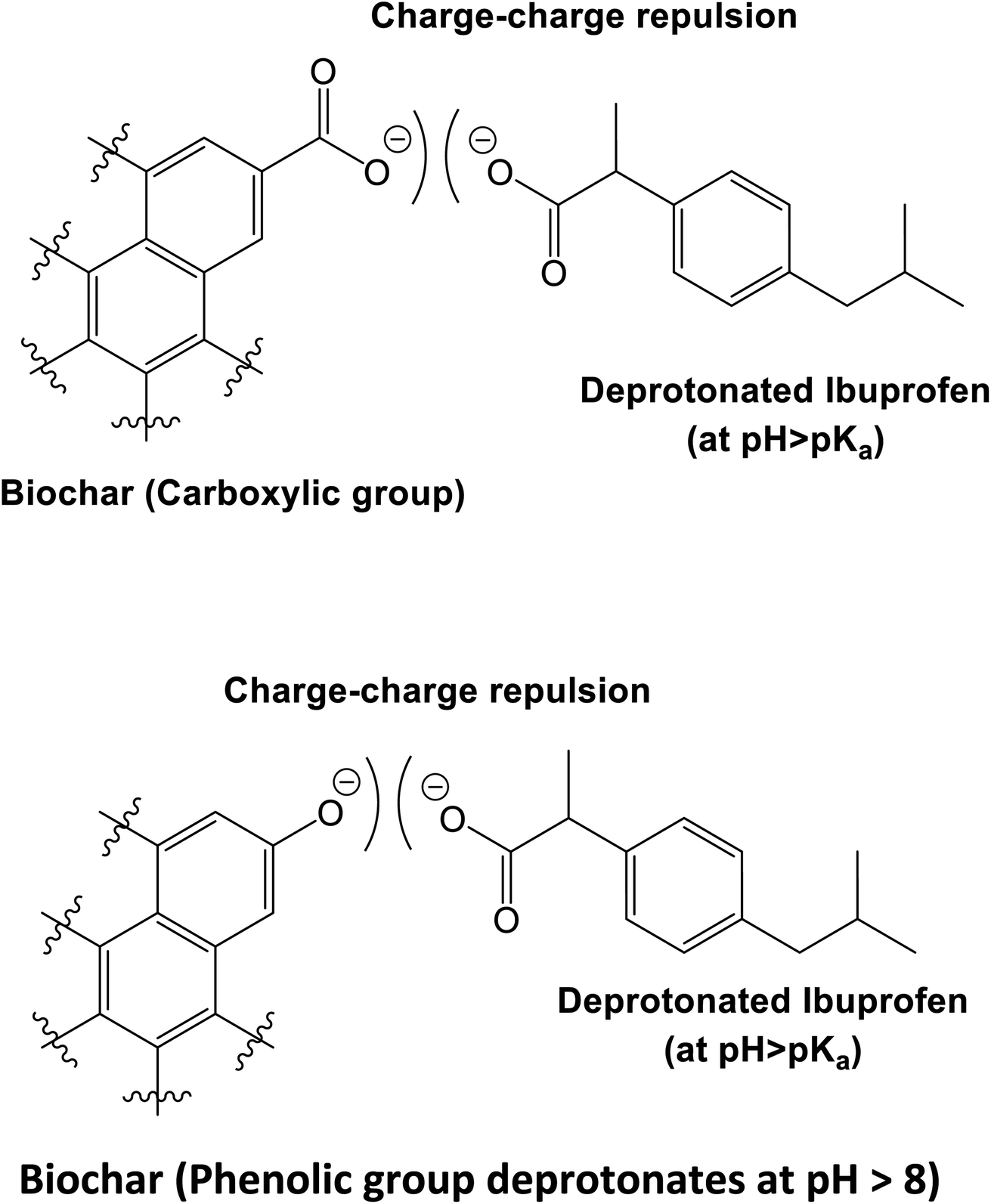
Aqueous ibuprofen sorption by using activated walnut shell biochar: process optimization and cost estimation - Environmental Science: Advances (RSC Publishing) DOI:10.1039/D2VA00015F

Multiple binding modes of ibuprofen in human serum albumin identified by absolute binding free energy calculations | bioRxiv

Universal Trends between Acid Dissociation Constants in Protic and Aprotic Solvents - Busch - 2022 - Chemistry – A European Journal - Wiley Online Library
![PDF] Sorption, photodegradation, and chemical transformation of naproxen and ibuprofen in soils and water. | Semantic Scholar PDF] Sorption, photodegradation, and chemical transformation of naproxen and ibuprofen in soils and water. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/bd825006414bdf48d3e25685d998bf571ff563c7/2-Figure1-1.png)
PDF] Sorption, photodegradation, and chemical transformation of naproxen and ibuprofen in soils and water. | Semantic Scholar

Comparative evaluation of ibuprofen co-crystals prepared by solvent evaporation and hot melt extrusion technology - ScienceDirect

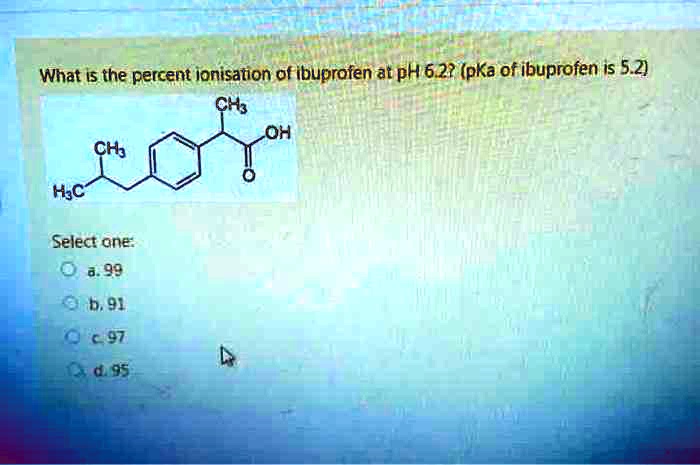
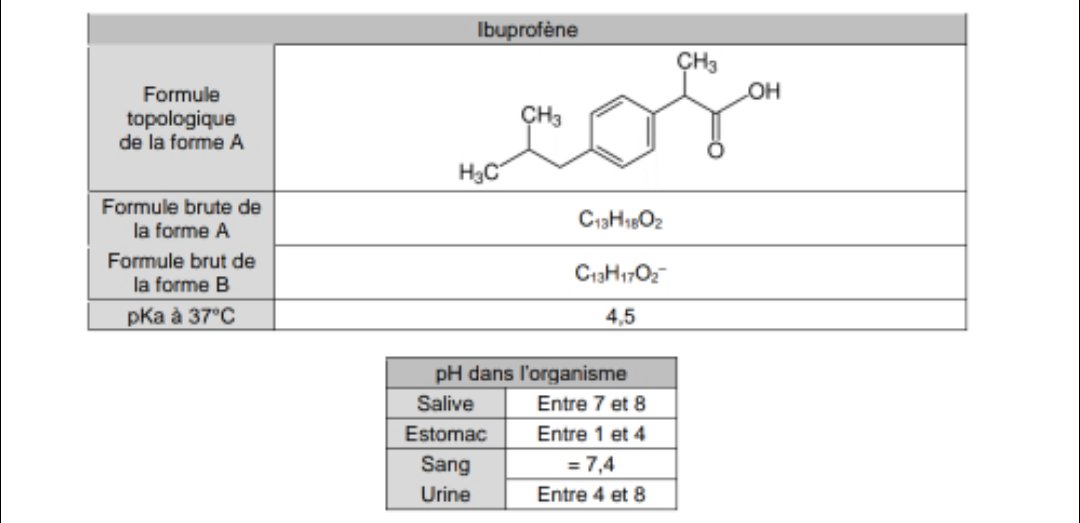
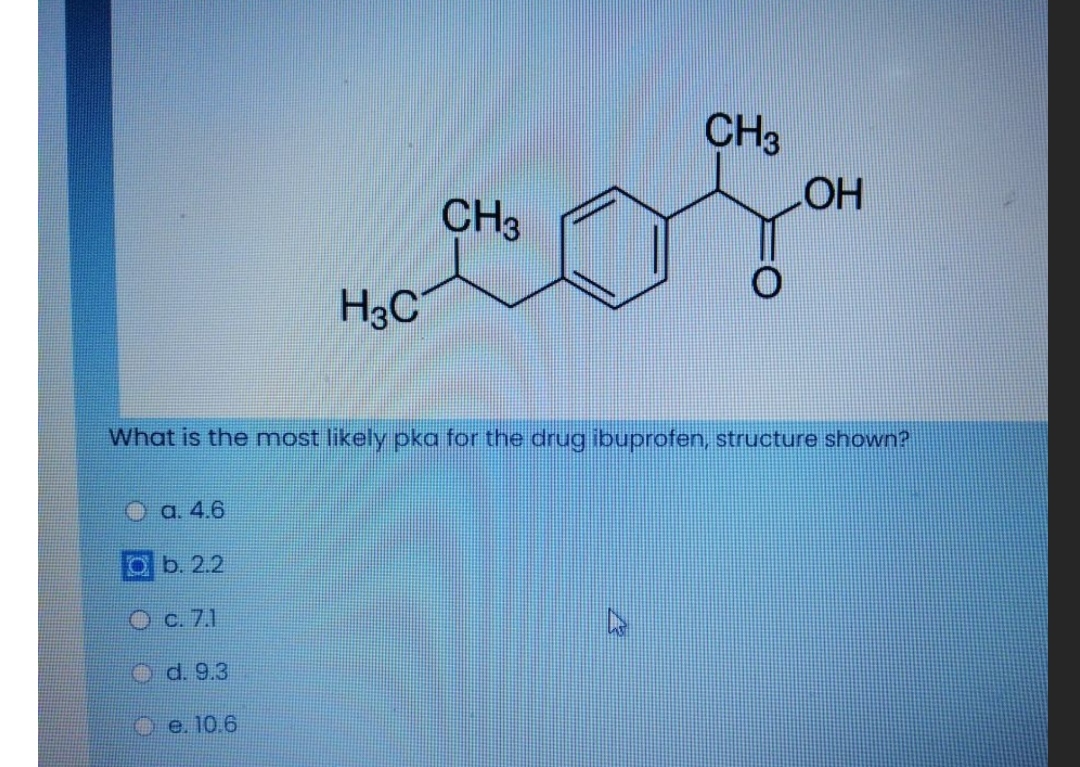
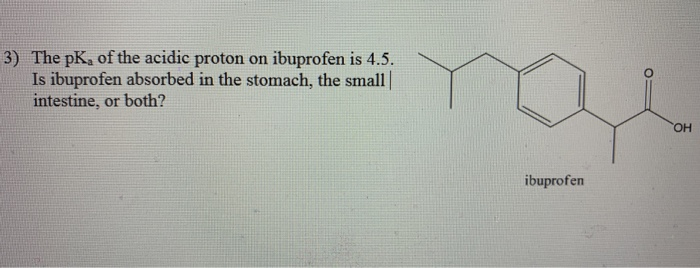
Ibuprofen (aka ADVIL) is a weak acid with a pKa of 4.9. It is absorbed through the stomach and the small - Brainly.com

Chemical structures and pKa values of the five different nonsteroidal... | Download Scientific Diagram
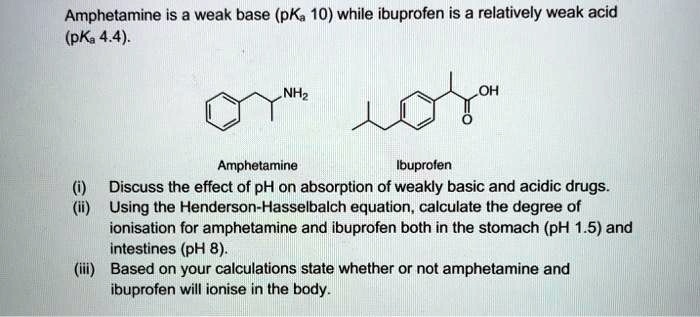
SOLVED: Amphetamine is a weak base (pKa 10) while ibuprofen is a relatively weak acid (pKa 4.4). Discuss the effect of pH on absorption of weakly basic and acidic drugs. Using the
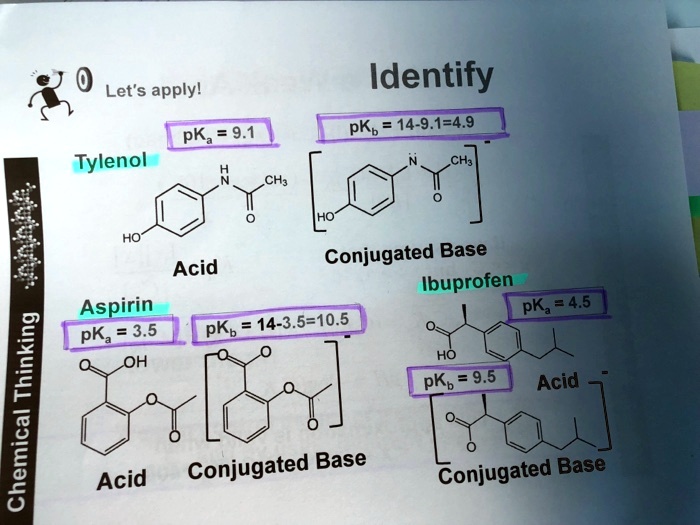
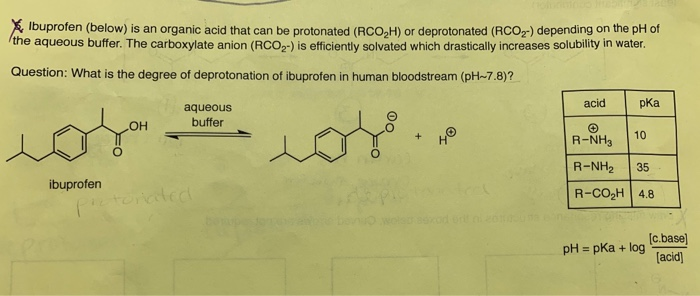
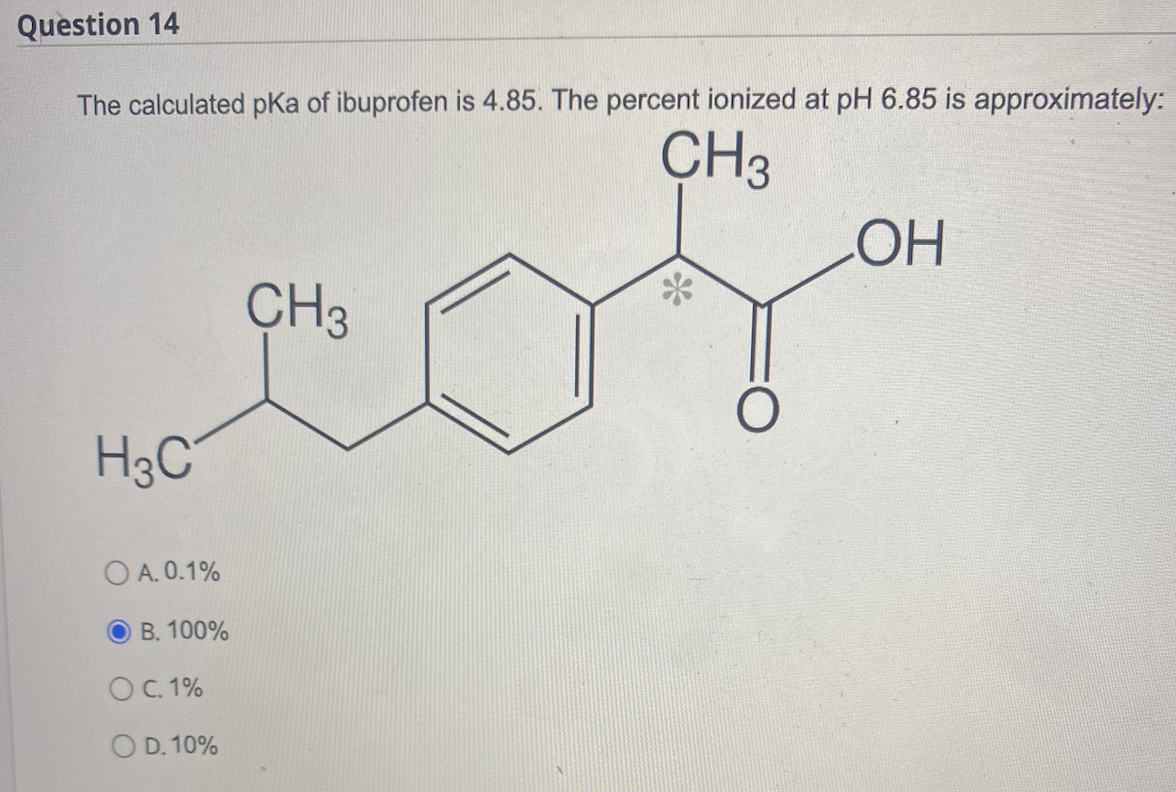
SOLVED: Let's applyl Identify pKb 14-9.1=4.9 pKa 9.1 Tylenol CHs CH; HO Conjugated Base Acid Ibuprofen Aspirin pKa =45 3.5 pKb = 14-3.5-10.5 pKa OH HO pKb 9.5 Acid 1 7 Acid Conjugated Base Conjugated Base
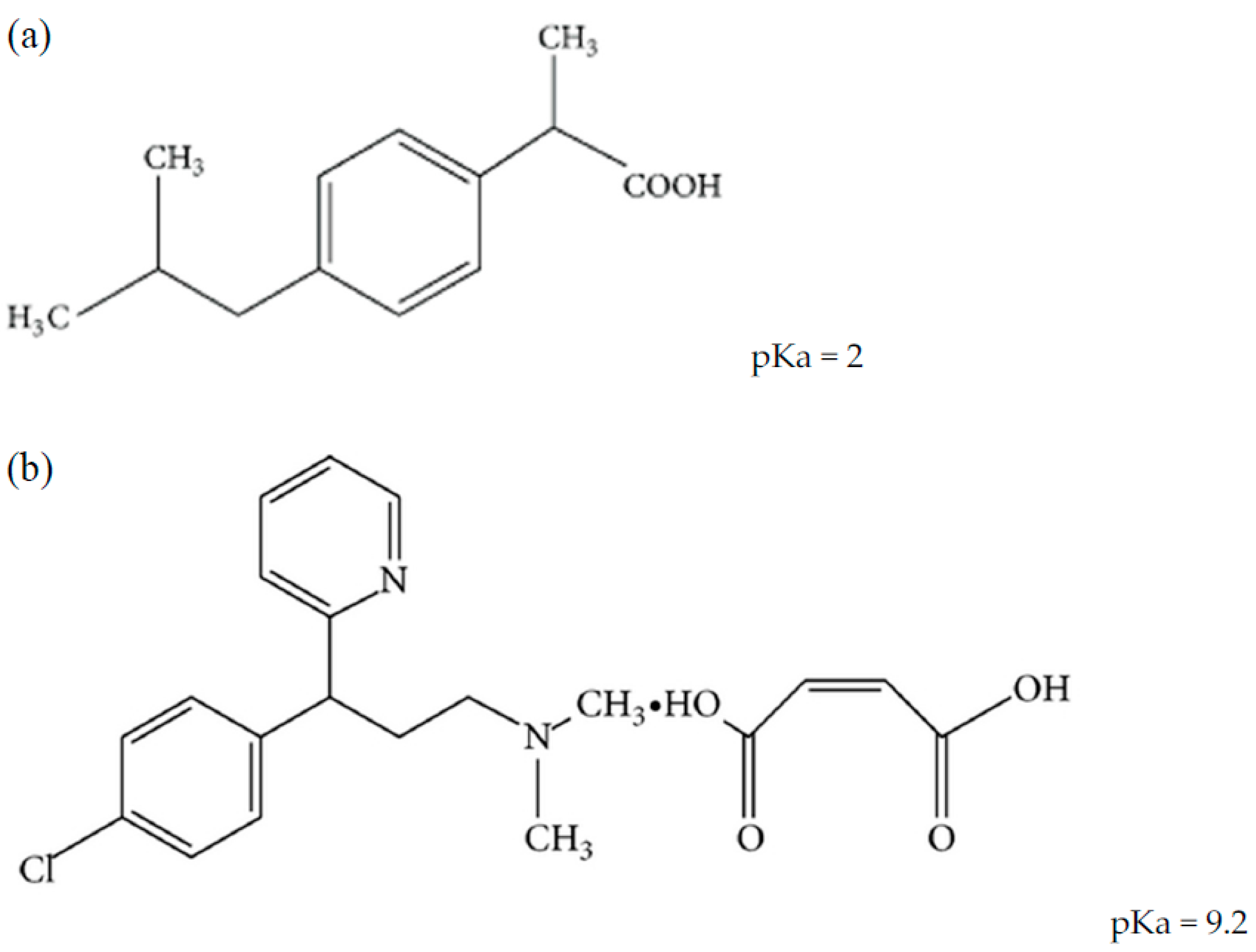
Sci. Pharm. | Free Full-Text | Development of HPLC Method for Simultaneous Determination of Ibuprofen and Chlorpheniramine Maleate

Ibuprofen: water affinity, effect of acidic pH and resonance structure:... | Download Scientific Diagram